2 PFAS Chemistry and Naming Conventions, History and Use of PFAS, and Sources of PFAS Releases to the Environment
The PFAS Team developed a Naming Conventions and Physical and Chemical Properties training video and a Production, Uses, Sources and Site Characterization training video with content related to this section.
This section provides a basis for the discussion of PFAS in the environment by presenting foundational information about its discovery, commercial use, health and environmental awareness, chemistry, and terminology.
In 1938, Roy J. Plunkett at the DuPont Company’s Jackson Laboratory discovered polytetrafluoroethylene (PTFE) while conducting commercial experiments with chlorofluorocarbon refrigerants (Science History Institute 2017).
Since the 1950s, many products commonly used by consumers and industry have been manufactured with or from PFAS, as the unique physical and chemical properties of PFAS impart oil, water, stain, and soil repellency, chemical and thermal stability, and/or friction reduction to a range of products. These products have applications in many industries, including the aerospace, semiconductor, medical, automotive, construction, electronics, and aviation industries, as well as in consumer products (such as carpets, clothing, furniture, outdoor equipment, food packaging), and firefighting applications (3M Company 1999; Buck et al. 2011; KEMI 2015; USEPA 2017).
The number of PFAS and their uses have expanded over the years. It has been estimated that the PFAS family may include more than 12,000 chemical substances (USEPA 2020). A recent inventory of PFAS identified more than 4,700 PFAS with Chemical Abstracts Service (CAS) Registry Numbers that could have been, or may be, on the global market (OECD 2021), although the uses of each of these PFAS may not be known (KEMI 2015). Publicly available health and toxicity studies are limited to only a small fraction of PFAS, and modern commercially available analytical technologies typically identify and quantify only about 50–60 PFAS, but these lists are increasing. An industry survey, reported in Buck et al. (2021), noted that only 256 of the 4,700 PFAS with CAS Registry Numbers are commercially relevant, with others of lesser environmental significance but potentially still occurring in the environment. However, it should be noted that (1) the results of this survey reflect responses from only three major global producers of PFAS that use the fluorotelomerization manufacturing process, (2) the results of this survey do not include any producers that use the electrochemical fluorination manufacturing process; and (3) some of the information provided by the participating companies was flagged as confidential business information.
Scientific, regulatory, and public concerns have emerged about potential health and environmental impacts associated with chemical production, product manufacture and use, and disposal of PFAS-containing wastes. These concerns have led to efforts to reduce the use of or replace certain PFAS, such as certain long-chain perfluoroalkyl carboxylates, long-chain perfluoroalkane sulfonates and their precursors, including two widely produced, commonly encountered, and most studied compounds: perfluorooctane sulfonic acid (PFOS) and perfluorooctanoic acid (PFOA) (USEPA 2016; WA DER 2017). Refer to the Water and Soil Regulatory and Guidance Values Table Excel file, the PFAS Regulatory Programs Summary Excel file, and Section 8 for updated regulatory actions, and Section 13 for stakeholder perspective.
PFAS have followed a similar pattern of emergence and awareness exhibited by many other anthropogenic environmental contaminants. Figure 2-1 provides a general timeline of PFAS emergence and awareness that includes categories of 1) synthesis/development, 2) commercial production, 3) health concerns, 4) environmental detection, and 5) reduction/alternatives. There may be evidence of some activities occurring before or after dates provided in this timeline; the intent of the figure is to capture the years of significance for each category.
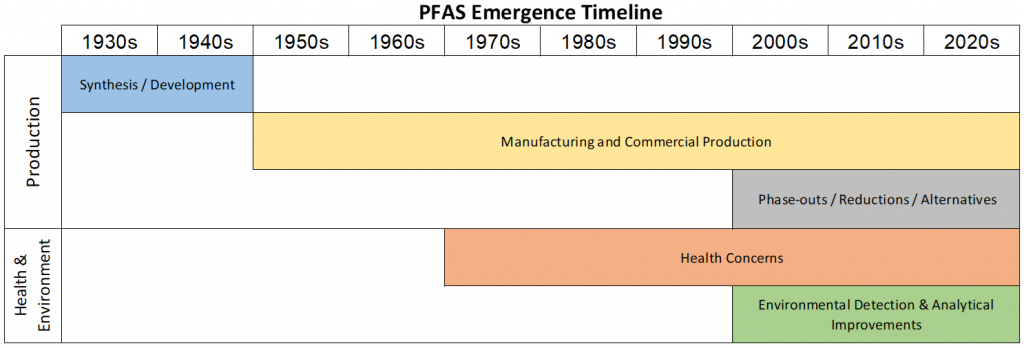 Figure 2-1. General timeline of PFAS emergence and awareness.
Figure 2-1. General timeline of PFAS emergence and awareness.
Graphic provides general indication of PFAS emergence and awareness by decade. Specific activities and events are described in more detail within this chapter. “Reductions/alternatives” refers to reduction in production/use and includes other PFAS that have replaced legacy chemistry.
Source: J. Hale, Kleinfelder. Used with permission.
The objective of this section is to lay a foundation for identifying potential PFAS sources in the environment.
Updated September 2023.